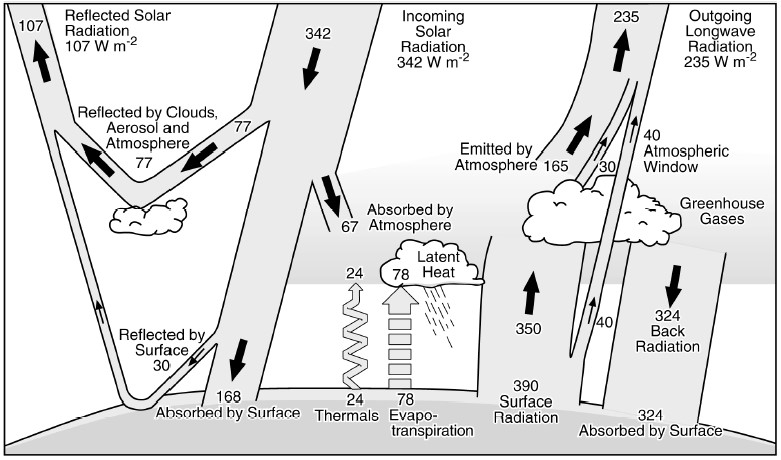
The greenhouse effect is the warming of the Earth due to its atmosphere. Solar radiation passes through the atmosphere like the panes of glass forming the roof and walls of a greenhouse. Radiant energy impinging on Earth’s surface and the floor of the greenhouse causes them to heat up. Since heat flows from hot to cold as a matter of basic physics, both of the now warmer surfaces heat the surrounding air by radiating upward. The greenhouse effect results because the solar radiation passes through the atmosphere and the glass with little absorption, but the surface heat radiation is partially absorbed as it seeks to escape. The reason for the difference is that the wavelengths of electromagnetic energy of the two are different. Solar radiation that reaches the Earth is shorter wave ultraviolet and visual. The heat radiation emanating outward from the surface is longer wave infrared. The terms ultraviolet and infrared refer to the wavelengths that are shorter than, or “beyond” the violet end of the visible spectrum and those that are longer than or “below” the red end (keeping ROY G BIV in mind). The significance of different wavelengths should come as no surprise. The microwaves used to heat up lunch while listening to the radio waves of music broadcast remotely are part of the same electromagnetic spectrum.
As diagrammed above, incoming solar radiation that reaches the top of the atmosphere is 342 watts per square meter (Wm-2 is shorthand for W/m2). Watt is the eponymous unit of power familiar from light bulb notoriety to honor James Watt, the inventor of the condensing steam engine. He coined the term horsepower so that people would understand what a steam engine could do; one horsepower is about 746 watts. Only 168 Wm-2 is absorbed by and heats the surface of the earth as 77 Wm-2 is reflected by clouds, aerosols, and atmospheric gases, 30 Wm-2 is reflected by the earth’s surface, and 67 Wm-2 is absorbed by the atmosphere. Thus, the sun’s primarily ultraviolet and visible light short wavelength incoming radiation mostly passes through the atmosphere, heating up the surface of the earth like it does a greenhouse. The outgoing surface radiation of 390 Wm-2 is shown on the bottom right. This is the longer wavelength infrared radiation of the Earth’s surface rising into the atmosphere. The change in wavelength between incoming and outgoing is because the sun is much hotter than the earth. [1]
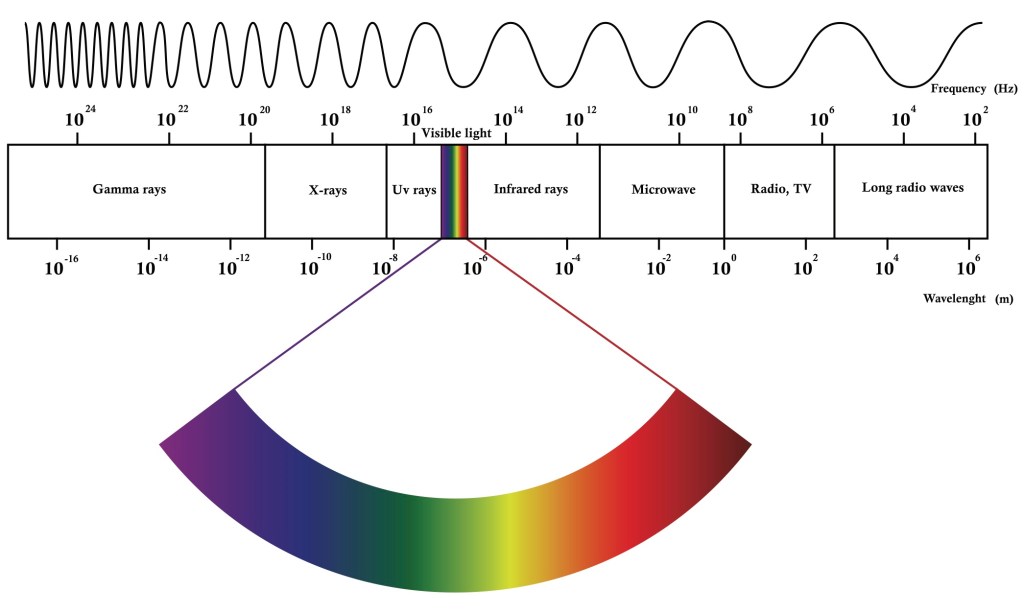
The radiation spread or spectrum between high energy ultraviolet and low energy infrared is based on the temperature of the radiating body. Some of the infrared radiation (40Wm-2) escapes, but over 80 percent (324 Wm-2) is reflected back to the surface by the gases in the atmosphere, which are called greenhouse gasses for this reason. The other heat energy components in the diagram are those associated with the hydrologic cycle; the evaporation and condensation of water is also a function of heat and temperature. The climate changing equation is that incoming short wavelength solar radiation must be either reflected back into space, or balanced by outgoing longwave radiation, mathematically 342 = 107 + 235. It is clear that the greenhouse gases play a key role in this balance. If more gas is added, more heat is radiated back from the atmosphere and surface temperature must go up to compensate. Global warming results. [2]
Trapping the heat of the sun under a warming blanket of atmosphere makes life on Earth possible. Without the greenhouse effect of its atmosphere, Earth would be like our planetary neighbor in the next outward orbital. Mars has an average temperature about 75°F below zero. If no action is taken to stem the tide of rising temperature, Earth will become more like Venus, where the mostly carbon dioxide atmosphere creates a super greenhouse effect with an average temperature of over 800°F. The planet hunting astronomers call the region near a star which falls in the range where liquid water can exist the Goldilocks Zone indicating that life as we know it could be possible there ― the circumstellar habitable zone. It is necessary but not sufficient that Earth is in one. It must also have sufficient moderating atmosphere with enough (but not too many) greenhouse gas molecules.
The French mathematician Jean-Baptiste Fourier is credited with making the first observation that the earth must be warmed by solar radiation due to atmospheric containment: “Tous les effects terrestres de la chaleur du soleil sont modifiés par l’imposition de l’atmosphère.” (all of the sun’s heat effects on earth are modified by the atmosphere). [3] This philosophical observation was rooted in science by the Swedish physicist Svante Arrhenius who first quantified the effect of carbon dioxide (then called carbonic acid) on temperature, which he called the “hothouse” effect (which, ironically, is probably the better term). His conclusion was “… if the quantity of carbonic acid increases in geometric progression, the augmentation of the temperature will increase nearly in arithmetic progression.” [4] The theoretical musings about the greenhouse effect became more factual after the first four decades of the 20th century. British engineer Guy Callendar reviewed historical data in 1938 to conclude that: “by fuel combustion man has added about 150,000 million tons of carbon dioxide to the air during the past half century” resulting in a measurable global temperature increase “at an average rate of 0.005°C per year.” [5] While convincing, the correlation of carbon dioxide to temperature does not prove causation – that the accumulation of atmospheric carbon dioxide is sine qua non for the measurable rise in global temperature.
The first scientific experiments were carried out by the Irish physicist John Tyndall who realized that the absorption of radiation by gases was “a perfectly unexplored field of inquiry” in 1859. He constructed the world’s first spectrophotometer, a tube that could be filled with different gases and subjected to radiation. It was instrumented with a recently invented device called a differential thermopile that could measure miniscule changes in temperature. Six months after he began his experiments, he presented his eureka results to Britain’s Royal Society: Different gases varied markedly in their ability to absorb and retransmit radiant heat. Nitrogen and oxygen, which make up over 99 percent of atmosphere were found to be essentially transparent to radiant heat, but other more complicated molecules, including water vapor, carbon dioxide, ozone, and (quixotically) perfume absorbed heat much more readily, even in small concentrations. Tyndall stressed the importance of water vapor, because “comparing a single atom of oxygen or nitrogen with a single atom of aqueous vapor, we may infer that the action of the latter is 16,000 times the action of the former.” [6] He concluded that water vapor was the most important gas controlling the surface temperature of the earth. This, then, became Royal Society gospel and accepted science for over a century.
The emergence of carbon dioxide as the true climate chimera was only a matter of time and science. Tyndall’s primitive experiment demonstrated only that humid air absorbed heat energy. Why it did so was another matter. The physics is complex, relating to the quantum energy levels of atoms of the greenhouse gas molecules. Spectroscopy, the study of the absorption and emission of light and other radiation as related to its wavelength, evolved rapidly in the early decades of the twentieth century. The emission or absorption of light within a narrow frequency and energy band is called a spectral line. Carbon dioxide has thousands of spectral lines that are responsible for the absorption of the infrared radiation of heat energy. A detailed understanding only became possible with accurate measurements at different heights in the atmosphere. These vary in intensity and width with temperature and pressure and therefore with altitude … a multivariable problem in three dimensions presenting a tangle of interrelated calculations. High speed computation was needed to run the iterative sequences of differential equations. By the 1950s, the measurements were available and the computers were programmed. The absorption of heat energy by molecules of carbon dioxide became settled cause and effect science. As early as 1956, there was convincing evidence that “…if the carbon dioxide content of the atmosphere should double, the surface temperature would rise by 3.6 degrees Celsius.” [7]
There remains the vexing problem of water vapor. There is a rational reason why water and its vapor loom large in debates about climate change causation. Weather, the fluctuating state of the atmosphere with elements of wind, rain, and sunshine that determines climate only when averaged over decades, is dominated by water. Rain in summer and snow in winter come from clouds that are condensed water vapor evaporated from liquid oceans, lakes, and rivers. Water is the most variable component of atmosphere and is central to climate variability and change. Oceans cover 70 percent of the Earth’s surface, contain over 96 percent of its water, produce 86 percent of all evaporation, and receive 78 percent of all rain. Spinning this sloshing volume at speeds of up to 1000 miles per hour between and around the embedded land mass continents of a tilted, heated globe produces weather.
Water vapor is a natural greenhouse gas. It is also the most heat absorbing of all greenhouse gases. The hydrologic cycle of evaporation, rain, and runoff has been going on for billions of years ― the planetary plumbing system. The storing of the sun’s heat energy as the latent heat of evaporation of water into the atmosphere (note figure above) and its release when the vapor condenses to fall as rainwater provides the energy for weather. To complicate matters, water vapor produces positive feedback. Warmer weather means more evaporation which increases the water vapor in the atmosphere which traps more heat which causes warmer weather. Positive water vapor feedback is considered the most important factor in amplifying the increase in surface temperature. Further, water vapor condenses into clouds, which are not gases, but contribute nonetheless to the greenhouse effect by absorbing and emitting the infrared heat radiation. But clouds also act a shield, cooling the climate by reflecting solar radiation. The variability of cloud formation and movement is one of the most profound conundrums of climate science. The only plausible way to address the chaotic interplay of sun, wind, and water was to develop increasingly sophisticated models that require high speed supercomputers. That has now evolved to many different models that can be compared and contrasted to narrow the uncertainty.
The Coupled Model Intercomparison Project (CMIP) was started in 1995 as a collaboration among models to compare results. First generation Atmosphere – Ocean General Circulation Models (AOGCM) used the physical dynamics of atmosphere, ocean, land, and sea ice as impacted by greenhouse gases and particulates called aerosols. State of the art Earth System Models (ESM) were more recently added to include the effects of biochemical carbon, sulfur, and ozone cycles. Model validation consists in part of inputting historical data to compare model output with the known result. The latest CMIP round was based on data collections that ended in 2013 to evaluate the relative efficacy of 56 different models from twelve countries including the United States, China, Russia, and Norway (where weather forecasting started). The conclusion was that doubling the amount of carbon dioxide in the atmosphere would result in a temperature increase of 2.1 to 4.7 degrees Centigrade. [8] It is worth noting that the 3.6 degree rise estimated in 1956 is consistent with this result. Modelling continues as carbon dioxide emissions and temperature keep rising.
Even though water vapor is the dominant greenhouse gas, it is essentially irrelevant to climate change just as it is paramount to weather. Its variability in the short term of weather is offset by its consistency over the long haul of climate. The rising concentrations of other atmospheric gases due not immediately impact weather ― but they are at the epicenter of the climate change problem because they have been and are being added to the atmosphere continuously. The conclusion made by the United Nations was that the only way to arrest climate change was to reduce the atmospheric emission of greenhouse gases over time. The Kyoto protocol was a United Nations (UN) treaty initiated in 1997 and which went into effect in 2005 after ratification by Russia and Canada as the last two of the stipulated fifty-five nation quorum. It specified limits on the six greenhouse gases that were found to be the most damaging due to heat absorption characteristics and concentration in the atmosphere: carbon dioxide (CO2), methane (CH4), nitrous oxide (N2O), sulfur hexafluoride (SF6), hydrofluorocarbons (HFCs), and perfluorocarbons (PFCs). The Global Warming Potential or GWP was established as a parameter with a molecule of carbon dioxide having a value of 1 in order to quantify the effects of the other gases. The UN Conference of Parties (COP) that constitute the signatories to the treaty agreed to proceed “with a view to reducing their overall emissions of such gases by at least 5 per cent below1990 levels in the commitment period 2008 to 2012.” [9,10]
The last three “minor” greenhouse gases are frequently grouped together as the “F-gases” to indicate that they contain the element fluorine; taken together, they constitute less that 1% of the total greenhouse gas emissions. Sulfur hexafluoride (SF6) gas is used primarily in high voltage electrical distribution systems due to its insulation properties. It was a replacement for oil-filled electrical components that contained polychlorinated biphenyls (PCBs) which were banned in 1979 by the Toxic Substances Control Act (TSCA). Each SF6 molecule is the equivalent (GWP) of 23,900 molecules of CO2. HFCs and PFCs consist of a number of different compounds that were formulated to replace chlorofluorocarbons (abbreviated as CFCs) that were banned by the Montreal Protocol of 1987 due to their ozone depleting effect (ozone filters damaging UV radiation). Their GWP values range between 140 and 11,700 for HFCs and between 6,500 and 9,200 for PFCs . In the 1950’s, Barry Commoner, a prescient scientist at the forefront of the environmental movement, devised four laws of ecology. [11] The irony of introducing greenhouse gases (HFC and PFC) to replace an ozone depleting substance (CFC) is direct evidence of his fourth law – “There is no free lunch” – every environmental solution (ozone depletion) has a cost (greenhouse gases). This applies equally to SF6 and PCBs.
Nitrous oxide (N2O) is the least known of the three “major” greenhouse gases, its provenance usually listed as “agricultural soil management.” With a GWP of about 300, it constitutes about 8% of the total greenhouse gas composition. The main culprit is fertilizer, which is about 10 percent nitrogen that must be added to the soil to compensate for the nitrogen removed with the harvest of the crop – about 100 pounds of nitrogen are removed with the harvest of every acre of corn. Fertilizer is necessary and sufficient to “manage” soil agricultural productivity. This added nitrogen is acted upon by the bacteria in the soil as a source of energy for their own growth and reproduction – a process that is called nitrification, basically the conversion of ammonium (NH3) into nitrate (NO3). Nitrous oxide is a naturally occurring by-product of bacterial nitrification of the added nitrogen-based fertilizer. Not to get too technical but to be complete, there is also a process called denitrification in anaerobic (lacking oxygen) soils where bacteria reduce nitrate to gaseous nitrogen; denitrification, like nitrification, releases nitrous oxide as a by-product. Thus, as more crops are grown for the ever-expanding global population for either food, fodder or fuel (ethanol), more nitrogen enriched fertilizer must be used to reconstitute the depleted soil – and therefore more nitrous oxide results. The Anthropocene nitrogen cycle has been called the Wibbly-Wobbly Circle of Life. [12] Commoner’s first law of ecology is “Everything is connected to everything else.” The earth is such a complex and balanced ecosystem that every disturbance (added fertilizer) has far-reaching effects (greenhouse gases and global warming).
The three primary sources of methane (CH4) with a GWP of around 30 are enteric fermentation, natural gas systems, and landfills. Taken together, they contribute more that three fourths of the total methane emissions in approximately equal shares. Enteric fermentation methane is from the normal digestion of food by ruminant animals, particularly cattle. Ruminants are named for the rumen, the first of their four stomachs – the repository for the fibrous material that they consume. Microbes in the rumen break down the tough cellulose as part of the digestive process; methane is a byproduct of that process that is expelled by the animal as exhalation. Over 95% of enteric fermentation methane is from beef and dairy cows. Other animals, including humans, produce the remainder of the enteric (intestinal) fermentation methane as flatulence. Methane is the primary constituent of natural gas that is widely used for heating and to generate electricity – some of this natural gas escapes into the atmosphere. Landfills are the largest of the three major sources of methane, comprising almost 40% of the total – the source is anaerobic bacterial decomposition of human trash. Commoner’s second law of ecology applies to methane – “Everything must go somewhere” – there is no way to simply throw things (trash) away, because it will still be there and you have to live with the results (greenhouse gases).
And last but certainly not least is carbon dioxide, the scion of the industrial age and perhaps the harbinger of its demise; it makes up more than 80% of all greenhouse gasses – by definition it has a GWP of 1. The carbon cycle is the essence of life; carbon dioxide is input to plant photosynthesis and output of organisms like humans oxidizing food for energy. The majority of excess carbon dioxide in the atmosphere comes from the combustion of fossil fuel – oil, gas and coal. It is the energy released by the oxidation of hydrocarbons that is both the boon and the bane of the modern world. For example, the natural gas reaction is:
CH4 + 2O2 ―> CO2 + H2O + energy
The level of CO2 in the atmosphere has historically been about 280 parts per million (ppm). It is now over 420 ppm. The energy we use to make electricity and to operate vehicles is increasing greenhouse gas concentrations which are causing the earth to heat up. “Nature knows best” is Commoner’s third law of ecology – every human made change is likely to be detrimental to the balance of nature. Anthropogenic greenhouse gases are the most obvious and potentially existential example. Our mother is nature.
References:
1. Third Annual IPCC Report – https://www.ipcc.ch/report/ar3/wg1
2. Dessler, A. and Parson, E. The Science and Politics of Global Climate Change, Cambridge University Press, New York, 2006, pp 6-11.
3. Fourier, J. “Remarques Generales sur les Temperatures Du Globe Terrestre et des Espaces Planetaires”. Annales de Chimie et de Physique. 1824 Volume 27 p 165.
4. Arrhenius, S. “On the influence of carbonic acid in the air upon the temperature of the ground” The London, Edinburgh, and Dublin Philosophical Magazine and Journal of Science. April 1896, Volume 41 No. 251: pp 237–276.
5. Callendar, G. “The artificial production of carbon dioxide and its influence on temperature” Quarterly Journal of the Royal Meteorological Society April 1938 Vol. 64 Issue 275 pp 223-240.
6. Fleming, J. Historical Perspectives on Climate Change, Oxford University Press, New York 2005. pp 66-74.
7. Plass G. “Carbon Dioxide and the Climate.” American Scientist, 1956, Volume 44 pp 302-316.
8. Flato, G. et al Evaluation of Climate Models. Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change Cambridge University Press, Cambridge, UK. pp 741-827.
9. Kyoto Protocol to the United Nations Framework Convention on Climate Change. Conference of the Parties. FCCC/CP/L.7/ADD.1, Kyoto, Japan, 10 December 1997.
10. https://www.epa.gov/enviro/greenhouse-gas-overview
11. Miller, Stephen. “Early Voice for Environment Warned About Radiation, Pollution”. The Wall Street Journal. Retrieved June 2018. In his 1971 best seller The Closing Circle, Commoner posited four laws of ecology: Everything is connected; Everything must go somewhere; Nature knows best; and There is no such thing as a free lunch.
12. Essay in The Economist, 24 December 2022.
